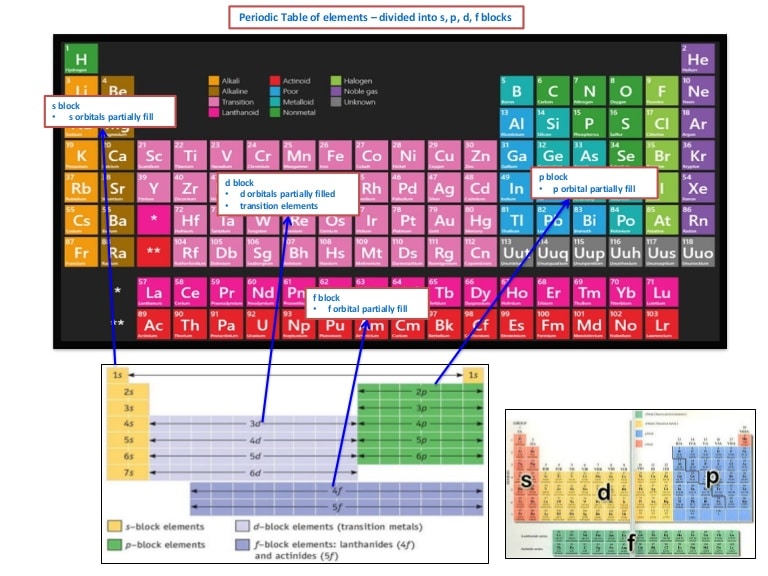
Periodic table with electron configuration The electron affinity trend of the second-row chemical element in the periodic table is usually lower than the third row. 
The lower member of crystalline solid metals like silver (Ag), gold (Au), cadmium (Cd), and mercury (Hg), the ionization energy increases.Įlectronegativity and electron affinity generally decreases but sometimes apparent some exceptions. The ionization energy usually decreases but in many cases increases due to shielding electron or effective nuclear charges. When we move down in a group, the size and electropositive character of atoms increases. In learning chemistry, we summarize physical and chemical properties along the group and period. Understanding the periodic variation of ionization energy, electron affinity, electronegativity, acid-base properties, and oxidation number in redox reaction of chemical elements are very important characteristics for any discussion or information in chemistry or science. The f-block on the periodic table is divided into two series, 4f or lanthanides and 5f or actinides which contain many missing elements discovered or synthesized by the nuclear reaction of radioactive isotopes. Therefore, 3d-block elements like scandium, titanium, vanadium, chromium, iron, cobalt, nickel, copper, and zinc are placed in the middle of the table between s and p-block with valence shell electronic configuration, 4s 0→2 3d 1-10. The transition or inner transition family forms the ionic chemical bond with metals (s-block) and covalent bond with metals non-metals (p-block). The name d-block (transition series) or f-block (inner transition series) on the periodic table is used due to the presence of progressively filled d or f-orbitals in the valence shell electronic structure. Since the second period of p block elements like boron, carbon, nitrogen, oxygen, fluorine, and argon have filled s-orbitals with valence shell electron configuration, 2s 2 2p 1→6, where n = number of period. Group-3, 4, 5, 6, 7, and noble gases belong to p-block elements. 
P-block on the periodic table organizes by progressively filled p-orbital in valence shell electronic structure but helium is an exception with electron arrangement 1s 2. The valence shell electron configuration, ns 1→2, where n = principle quantum number, or the number of periods. Group-1 (hydrogen, lithium, sodium, potassium, rubidium, cesium, and francium) and group-2 (beryllium, magnesium, calcium, strontium, barium, and radium) belong to s-block elements. The name s-block element in the periodic table is given due to the arrangement of electrons, the valence electron enters into ns-orbital and is filled progressively according to the configuration rules. The organization of the middle portion of the periodic table made the relationship between the left and right sections and contains a list of transition metals or d-block and inner transition or f-block elements.Īccording to the modern periodic law based on the atomic number and valence shell electron arrangement of elements, the different types of metals and non-metals in chemistry are organized to form s, p, d, and f-block on the periodic table.The right portion contains groups IIIA, IVA, VA, VIA, VIIA with high electron affinity, and non-reactive noble gases like helium, neon, argon, krypton, xenon, and radon.The left section in the table has group IA and IIA define the extremely high electropositive character but very low ionization energy and always show the positive oxidation number.According to the above definition and explanation, the long form of the periodic table is labeled into three units like left, right, and middle sections for learning chemistry or physics in science.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
